Category: "Ice Science"
The Curious World of Ice and Snow: Part 2 of 3
February 8th, 2020As I mention in Part 1, these are slides I gave for a Science Cafe discussion session in 2012. This section focuses on ice that forms directly from the melt, that is, the liquid phase. Contrast these cases with those in part 1 in which the ice grew from the vapor. Some of these cases might seem a little familiar, but many ought to seem downright bizarre.
As always, click on an image to enlarge it.
The contents here are emphasized in green font below. The underlying difference between ice growth from vapor and from melt is that the melt is much denser. The higher density means that vastly more water molecules can impinge on the ice surface in a given time and area. This higher impingement usually means faster growth and larger crystals, but the way that the melt reaches the crystal influences the form, and the result is not always so obvious. In fact, I would even say that it is never obvious.
OK then, let's get right back into it.
The Curious World of Ice and Snow: Part 1 of 3
February 4th, 2020In 2012, I gave a "science cafe" talk with a local series sponsored by the Pacific Science Center, KCTS public television, and Science on Tap. The title was "The curious world of ice and snow". The location was a bar in Kirkland, but open to all ages. When I showed up with my family, they tried to seat us in the backup room, the regular room having filled up, but I said "Oh, well I'm the speaker" and they kindly created a space for my family in the regular room. I was indeed surprised at the crowd. People are apparently more interested in ice than I thought. (Hmm, but where are they when I post here?)
Click on any image to see an enlargement.
The basic structure of each talk was to give a lecture of about 30 minutes and then allow up to an hour (I think) for the Q&A. In my excitement, I had created 41 slides, in retrospect too many for the allotted time.
Given all the time spent preparing the slides, I hope that by posting them here that even more folks can enjoy the images and discussions. But, instead of unloading all of them on you at once, I will break the discussion into three sections. By adding the following table of contents, each section will have 14 new slides and the total will be 42, which according to Douglas Adams* is a really special number.
The contents of this section is part "1", written in green font.
The Growing Icicle's Hollow Tip
January 17th, 2020If you inspect the tip of a growing icicle, you might be surprised to find it hollow. Skeptical? Well, if you think the conditions suitable for icicles outside, put a toothpick in your pocket and go outside. Then when you see a likely candidate, poke its tip with the toothpick.
Click on the image to enlarge.
Not all icicles are growing, of course. If the supply of flowing water has dried up, the tip may be solid, or if the air has gotten warm, then the tip may be rounded and wet. The pic below shows a longer, but solid-tipped, icicle next to the growing one. Its melt-water source is too low for the tip to grow.
Transitional situations occur as well, such as the icicle that just started to melt and still has a hollow tip, or the completely solid icicle that just starts growing again and has yet to develop a discernable hollow. The drip may also be intermittent. You might not find that growing icicle right away, but keep that toothpick handy.
As to why a growing tip is hollow, I won't attempt to prove it here, just try to make it seem plausible. Spend a few moments considering the following two simpler situations that show the basic ideas. You can even apply these ideas to other forms involving freezing water...
Martini Hoar (raise a tiny glass?)
October 19th, 2019The hoar-frost crystal shoots up like a thin, solid straw, then suddenly opens up into a cup-like shape. I have seen it often enough to give it a name: "martini hoar".
The cup can be weirdly segmented and polyhedral, but it nevertheless widens suddenly. Here are a few more (Sorry for poor photos—someday, I hope, I'll get better about photography.)
Here is a larger view of the region. Note the similar hoar coming down from the top, but without a clear view of the base.
This sudden widening feature has bothered me for awhile, but I was delighted the other day to figure out a plausible reason. My delight was made even greater because the reason involved measurements I made in the lab two decades back. The measurements were to understand snow-crystal habit, but apply equally well to hoar frost because hoar grows just like snow except it is attached to the ground.
Now that I have viewed some of the older pictures I took, the actual growth phenomenon looks more complicated in terms of crystal shape, so I am not so sure my reasoning explains things so simply. Nevertheless, it should apply well to many cases, and at least is worth learning because it involves important growth principles that also apply to snow.
Puddle gets its grooves (upon freezing), part I
February 16th, 2019These grooves appear on the surface of frozen puddles.
Most grooves are straight lines, and most of these also appear to have relatively symmetric sides, such as those marked #s 1, 2, & 4 in the above image. But some, such as #3, have sides that are much wider. Grooves can intersect each other at a "T" intersection, such as #1 and #3, or they can cross, as a 4-way intersection as in #2. Some, such as #4 start and end on nothing apparent and intersect no other grooves.
Sometimes the sides show small steps or sub-grooves, particularly on the wider sides, such as on #3. On grooves that curve, such as in #1 and #2 in the image below, one side can have a fern or foliage type of texture.
Grain boundaries between crystals in big ice
November 6th, 2017Most snow crystals are single crystals. Being single is an outcome of their growth process and small size. On the other hand, most larger ice formations are not single crystals. These latter types are called "polycrystals". A polycrystal usually appears the same as any other type of ice -- smooth, uniform, clear or white -- just as if it were also single crystal. But the poly nature can be revealed when the ice warms to the melting temperature. At melting temperature, the boundaries between the separate crystals become visible. As an example, note the white lines in the ice in the inset in the image below.
(This image is from the ice drip complex in the previous post.) We call each individual crystal a "grain", and the boundaries between grains as "grain boundaries". The grain boundaries show up because the region has disordered ice that will melt at a lower temperature than regular ice. Thus, the light, upon passing through a grain boundary, will scatter more, making a whiter region, as shown in the image.
The ice is thus weaker on a grain boundary and has a tendency to break along these boundaries. On the surface of ponds and lakes, the grains (individual crystals) can be several inches or more across. So, if you get a sheet of such ice, and let it warm up to melting, you will find it easy to break the ice sheet along a grain boundary and thus isolate a large single crystal.
The size and pattern of the grains affect the mechanical properties of the ice. So, glaciologists, who want to know how a given glacier or ice sheet moves, are very interested in such patterns. They call this pattern the "ice fabric".
-JN
How clouds form snow
January 14th, 2017To understand snow formation, one must know a little about clouds.
Q: What is in a cloud?
A: Air, dust, vapor, droplets, and often, ice.
Q: How much air? How much liquid water? How much ice?
A: The answers will probably surprise you. See my short 20-min presentation below. I gave this recently to the Bellingham, WA Snow School. (23 slides, but due to file-upload-size restrictions, I had to put them into three parts below, 10 slides, 6 slides, 7 slides.)
Snow, rain, and weather affect everybody, yet how many of us learned in school even the most basic facts about precipitation in school?
Q: Who first realized how ice grew in a cloud?
As described in my presentation, he realized this by observing frost on the ground.
Q: Who first realized how Alfred's theory was intimately connected with rainfall?
Tor discovered this by observing fog in a mountain forest, and like Alfred, applied some of his physics knowledge.
In my presentation, I discussed Alfred Wegener, the roles of the different cloud components, and briefly how the ice, once formed, takes on its strange shapes:
First 10 slides (with blue text added to account for the things I said during the talk):
http://www.storyofsnow.com/media/blogs/a/Jan2017/snowschool_annotated1t10.pdf?mtime=1484585328
Next 6 slides:
http://www.storyofsnow.com/media/blogs/a/Jan2017/snowschool_annotated11t16.pdf?mtime=1484585328
Last 7 slides:
http://www.storyofsnow.com/media/blogs/a/Jan2017/snowschool_annotated17t23.pdf?mtime=1484585309
Later, I will show specifically how the ice gets arranged into all these strange shapes.
- JN
Lots of Hair Ice
December 19th, 2014My older sister sent me this photo on a recent morning.
This is hair ice growing from an alder tree. All of it grew overnight, formed when liquid water near the outer trunk surface (beneath the bark) froze. This creates an ice front in each pore-like structure. Water inside the trunk flowed out to the ice front, pushing each hair-like strand of ice outward.
It probably does not get as cold under bark unless the bark is partly peeled off, so the hair ice comes out of bare spots in the trunk, as shown in the photo above. The air temperature got down to 26 F that night, and the tree was next to a dirt road, and thus partly exposed to the sky.
Like the needle ice that grows out of the ground, it grows relatively fast, producing relatively long hairs. From another spot, down near a meadow:
--JN
Glaze-rime Ice Buildup Facing Creek
November 23rd, 2014Here's a reed of some sort, with a glaze of clear rime on the side facing a creek. How did the clear ice get there, and what is the white ice on top of it?
The fact that the clear ice is just on the side facing the creek indicates that the ice came from droplets blown off the creek. To have the ice buildup just on the side means that the droplets must have frozen soon after landing, but slow enough to spread out and fill gaps.
So, the droplet must have been larger than typical cloud droplets, and the temperature must have been within a few degrees of freezing. Clearly, the flowing water must have been above freezing, so the droplets must have cooled to below zero while drifting through the air.
A closer shot shows that the outermost ice, the white ice, is hoarfrost. Only hoarfrost would show the flat crystalline facets. Definitive proof that that ice grew out of the vapor that was blowing by. You can also see some hoar on the other side of the reed.
--JN
Smaller Hoar Growing on Larger Hoar
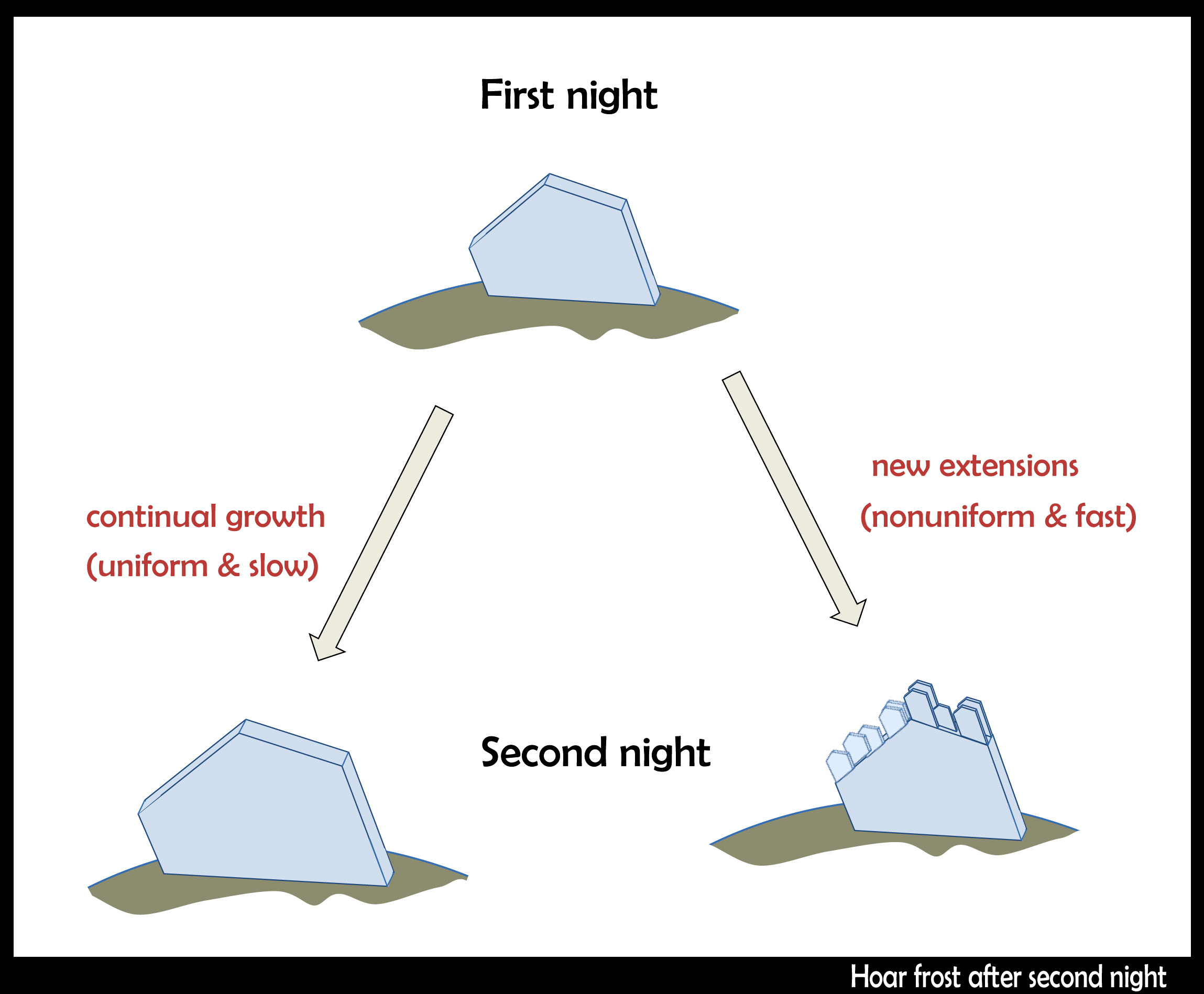
November 22nd, 2014Cold air came down the interior, spilling through gaps in the Cascades, cooling the western side. In the Seattle area, we got our first frost on Nov. 11, but the days stayed cold. Areas in the shade never lost their hoar, so the hoar frost kept growing.
But does a hoar-frost crystal continue growing the same way as on the previous night? Does it keep extending uniformly, getting larger and larger, keeping the same general shape?
Or do smaller hoar crystals form on previously grown larger crystals?
A few days later, I found myself walking in Maple Falls near Mt. Baker. In a shady pocket near my feet, I saw some hoar. Subtly different -- it appeared like small, translucent leaves encrusted on their edges with hoarfrost. But it turned out that the "small leaves" were actually tabular hoar (plate-shaped). That is, hoar encrusted on hoar: smaller crystals on larger crystals.
When frost resumes growing on previously grown frost, the other possibility may also happen; that is, the hoar just resumes growing the way it did the previous night. But I think this is generally quite rare because it would require very slow and constant growth conditions, which is generally incompatible with the diurnal temperature cycle. See the sketch below for both possibilities.
Note that I write above 'smaller hoar crystals grow on the larger ones' even though technically, they are all the same crystal. You can see this is true by the fact that their faces all line up.
The view that got me to look closer:
- JN